General Overview: Acute Gastroenteritis (GE) is one of the most common infectious diseases of all age groups worldwide, mainly in the pediatric population and it is a leading global cause of morbidity and mortality among children under 5 years of age in developing countries where complications include electrolyte disturbances and malnutrition that necessitates hospitalization [1]. The condition can be caused by some different microorganisms, but viruses are increasingly recognized as predominant causative factors. To note that acute GE is usually acquired by fecal-oral route, also through contaminated surfaces or from water sources. Identified symptoms vary between watery diarrhea, nausea, vomiting, fever and abdominal pain [1].However, viruses have become more frequently identified mainly with the advances in the ability to diagnose viral infections. Among them, Rotavirus (RV) is the leading cause of acute GE in infants and young children worldwide [1]. Although the first human RV was discovered in 1973; each year, about 25 million episodes of GE due to RV are reported in children worldwide, of which 2 million require hospitalizations and 180000-450,000 deaths occur less than 5 years of age, mostly in developing countries. In Lebanon, prevalence of RV infection has been estimated between 27.7 and 30.6% [1] (Figure 1).
The spectrum of RV illness ranges from mild, watery diarrhea of limited duration to severe diarrhea with vomiting and fever. The dehydration that can result can be more serious than the infection itself and is the most common cause of death in rotavirus cases [1]. Because the clinical features of RV related GE are similar to other viral and bacterial GE, confirmation of RV infection by laboratory testing of fecal specimens is necessary. The RV is shed in high concentration in the stool of children with gastroenteritis (i.e., 1012 viruses/G), so the best inexpensive and available method is antigen detection in the stool by an Enzyme Immunoassay (EIA) directed at an antigen common to all rotaviruses. Also other techniques can be cited but mainly used in research settings ex: electron microscopy, reverse transcription-polymerase chain reaction RT-PCR, nucleic acid hybridization, sequence analysis and culture [2]. Since 2006, two rotavirus vaccines are currently licensed for infants worldwide for routine immunization and have been introduced in both developed and developing countries leading to herd immunity in several states. So since RV vaccines were implemented, studies have been undertaken to assess the effects of vaccination on RV disease [3]. This study will discuss the impact of the RV vaccination program on relative reductions of RV disease burden and RV related GE hospitalizations among Lebanese infants and children aged less than 5 years in 3 different hospitals, using file or electronic records for data collection.
Review of literature
Epidemiology: RV remains the world’s commonest cause of GE among children predominantly in developing countries; globally RV alone is responsible for almost 40% of GE related hospitalization. In developing countries, Rotavirus is responsible for around half a million deaths annually. In addition, diarrhea caused by RV infection plays a major role in malnutrition among children and predisposes them to other infectious diseases which lead to increase in childhood morbidity and mortality. It is prevalent in all geographical regions of the world and almost all socioeconomic groups. Three of the seven RV groups (A to G) are known to infect humans. Among them, the most dominant is group A, which causes diarrheal diseases worldwide.
Rotavirus structure: The name RV derives from the characteristic wheel-like appearance of the virus when observed by electron microscopy, which is derived from the Latin word “Rota” meaning “Wheel”. This pathogen was initially discovered in diarrheic cattle, mice and monkeys and finally in infants and young children in 1973 by Bishop and Flewett [4]. RV is a triple layered, non-enveloped virus within the family of Reoviridae. The genome of this virus consists of 11 double-stranded RNA segments. The nucleic acid is surrounded by 2 layers of capsid–inner capsid (VP6) and outer capsid (VP7). In addition to VP4, the spike protein, this is a cell surface receptor [5]. This 11 segmented double-stranded RNA (dsRNA) encoding six structural Proteins (VP) and 6 Non-Structural Proteins (NSP). The viral proteins VP1, VP2, VP3, VP4, VP6 and VP7 are, as we mentioned, the structural proteins that form the virion. The non-structural proteins (produced by the cell infected by rotavirus) are NSP1, NSP2, NSP3, NSP4, NSP5 and NSP6 [5] (Figure 2 and 3).
Rotavirus Genotype
RV is classified into P and G serotypes, respectively. The genetic diversity of the virus is due to the segmented nature of its genome. Of the 11 genomic segments of RV, segment 6 codes for the most abundant viral protein, the VP6 protein, this is the major antigenic determinant of group A reactivity. Also, based on the VP-6 capsid gene, the virus has been classified into the seven major genogroups A to G. The Segments 7, 8 or 9 codes for the major glycoprotein, VP7, which is the basis for RV serotyping. Additionally, Segment 4 codes for a protease-sensitive protein, VP4, which is the basis for P-serotyping. So based on the sequence of VP7 genes (segment 9 mainly) and VP4 genes (segment 4), RVAs are classified into G and P-genotypes, respectively. Currently, there are 14 G serotypes and 11 of them have been detected in humans. The majority of human RVs belong to serotypes G1–G4 and to the newly emerged G9. Among the P types, P[8] was the most common type found across all G types [6]. To note that the genotype combinations which causing 90% infections in humans are G1P[8], G2P[4], G3P[8], G4P[8], G9P[8] and G12P[8] [7]. So based on that, the two live-attenuated oral rotavirus vaccines licensed for infants <6 months old contains human G1P[8] serotype for Rotarix while RotaTeq contains a mixture of five human serotypes G1–G4 and P[8] [7].
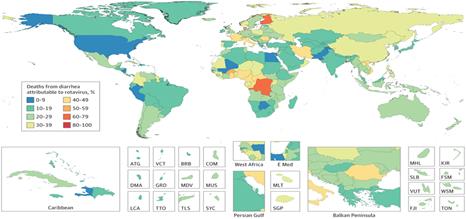
Figure 1: Rotavirus Related Mortality Worldwide. Adapted and Reprinted from Troeger C, Khalil IA, Rao PC, Cao S, Blacker BF, Ahmed T, Armah G, Bines JE, Brewer TG, Colombara DV, Kang G, Kirkpatrick BD, Kirkwood CD, Mwenda JM, Parashar UD, Petri WA Jr, Riddle MS, Steele AD, Thompson RL, Walson JL, Sanders JW, Mokdad AH, Murray CJL, Hay SI, Reiner RC Jr. Rotavirus Vaccination and the Global Burden of Rotavirus Diarrhea Among Children Younger Than 5 Years. JAMA Pediatr. 2018 Oct 1; 172 (10):958-965. Copyright © 2018 Troeger et al. JAMA Pediatrics
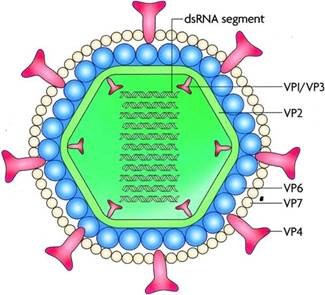
Figure 2: Rotavirus Structure. The Genetic Material is found inside a Complex 70-Nanometre Viral Nucleocapsid with three Concentric Shells: An Inner Core, an Internal Capsid vp6 and an Outer Capsid vp7. Sixty Spikes vp4 between 10 and 12 nm in Length Protrude from the outer Capsid. Adapted and Reprinted from Shaheen MNF. Rotavirus Gastroenteritis among Hospitalized Children under 5 years of age in the Eastern Mediterranean Region: A Review. East Mediterr Health J. 2019 Aug 19; 25(6):422-430. Copyright © World Health Organization (WHO) 2019
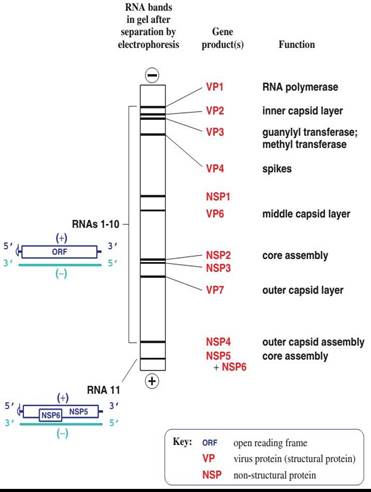
Figure 3: 11 Segments of the Genome of Rotavirus. Adapted and Reprinted from Shaheen MNF. Rotavirus Gastroenteritis among Hospitalized Children under 5 Years of Age in the Eastern Mediterranean Region: A Review. East Mediterr Health J. 2019 Aug 19; 25(6):422-430. Copyright © World Health Organization (WHO) 2019
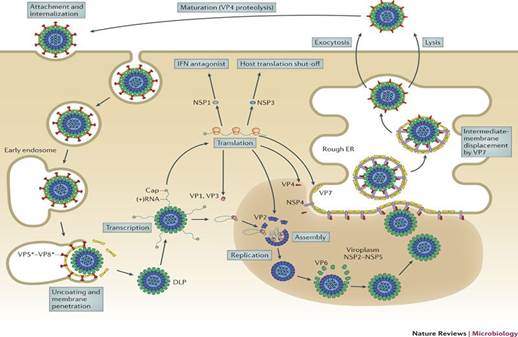
Figure 4: Replication Cycle of Rotavirus. Adapted and Reprinted from Online Biology Notes. Guarab Karki. Rotavirus-Classification, Structure composition and Properties, Replication, Mode of Transmission, Pathogenesis, Clinical Symptoms, Laboratory Diagnosis, Treatment, Prevention and Control. Available from: Rotavirus-Classification, Structure Composition and Properties, Replication, Mode of Transmission, Pathogenesis, Clinical Symptoms, Laboratory Diagnosis, Treatment, Prevention and Control - Online Biology Notes
Replication Cycle of Rotavirus in a Host Cell
The proteins VP4 and VP7, which compose the most outer layer, are important for the first interactions of the virus with the host cell surface by the attachment of the VP4 to the cell surface receptor. Rotavirus enters cells by endocytosis mediated by this receptor. During this entry, uncoating occurs in lysosomes leading to a Double Layer Particle (DLP) from the initial Triple Layer Particle (TLP) by losing the external protein layer. The transcriptionally active DLP starts transcription of the viral genome and the process is mediated by endogenous virus dependent RNA polymerase (transcriptase). The viral mRNAs direct the production of the viral structural and non-structural proteins by translation of this viral transcript and also it serves as RNA templates for the synthesis of negative sense RNA strand with capsid to form the double stranded RNA (dsRNA). The new viral proteins and RNAs accumulate in protected sites of the cytoplasm called viroplasms, where the viral genome replicates an double-layer intermediate replication particles assemble and form an inner capsid. The interaction of newly formed polymerase complexes with the core capsid protein triggers genome replication, which is followed by addition of the intermediate protein layer of the virion. These newly formed DLPs start budding into Rough Endoplasmic Reticulum (RER) and acquiring a pseudo envelope followed by subsequent removal of this transient lipid envelope which will be replaced by an outer capsid in RER to finally yield mature infectious TLPs. After release of TLPs through cell lysis or trafficking, the attachment spike must be cleaved by trypsin-like proteases in the intestinal lumen to activate the virus for subsequent infection [8] (Figure 4).
Transmission of Rotavirus
The primarily documented mode of RV transmission is fecal-oral route, directly from person to person, or indirectly via contaminated fomites or surfaces. A respiratory mode of transmission has also been proposed but it is not the usual mode of transmission [9]. In addition, Community wide water borne epidemics of group A RV (G-A RV) have been documented in Turkey in 2011 [10]. The young infant can get infected either from older siblings or patients with subclinical infection. And as known, Rotaviruses are highly contagious and a low viral load may trigger a human infection (<100 virus particles is enough to cause infection) [11].
Incubation Period
The primarily documented mode of Rotavirus transmission is faeco-oral route, directly from person to person, or indirectly via contaminated fomites or surfaces. A respiratory mode of transmission has also been proposed but it is not the usual mode of transmission [9]. In addition, Community wide water borne epidemics of group A Rotavirus (G-A RV) have been documented in Turkey in 2011 [10]. The young infant can get infected either from older siblings or patients with subclinical infection. And as known, Rotaviruses are highly contagious and need low doses for human infection (<100 virus particles is enough to cause infection) [11]).
Distribution
RV is distributed throughout the world and they are the major causative agent of severe diarrhea with dehydration in infants and young children. According to WHO reports, there are more than 25 million outpatient visits and more than 2 million hospitalizations that were attributed to RV infections. It also estimated that 82% of global Rota viral deaths occur in poorest countries such as Africa, Asia and Latin America [9].
Seasonal Variation
Before the introduction of vaccination, RV showed marked seasonal variation in developed countries with the epidemic peaks occurring in the cooler months of the year, but the seasonal peaks of the infections can vary broadly and occur from autumn to spring. Following the introduction of Rotavirus vaccination, the seasonal pattern has decreased markedly. On the other hand, in the tropics, less or minimal seasonal variations have been noted even before vaccination. This may be due to high birth rates as well as high transmission rates found in developing countries when many of these countries lie in the tropical belt [12] (Figure 5).
Risk Factors
The Age has been identified as an important risk factor for RV infection: In developed countries, RV GE mainly affects infants and young children between 6 months to 2 years of age followed by the infants younger than 6 month of age and it is generally asymptomatic in neonates due to maternal antibodies which are transferred transplacentally during pregnancy. Concerning the Sex, male children were found to be two times more susceptible and more hospitalized than females [13,14] (Figure 6).
For the other risk factors, such as Socioeconomic Status (SES), the association is less clear, with certain results observed among studies that reported higher rates of RV infections among lower socioeconomic groups because of poverty, suboptimal living conditions and low uptake of the optional vaccines [15]. In addition, illiteracy, low birth weight, inadequate breastfeeding, malnutrition is also linked with higher incidence.
Pathogenesis
The pathogenesis of Rotavirus is multifactorial and incompletely understood. The virion infects villous enterocytes and causes cell destruction. The possible role of Rotavirus in pathogenesis of gastroenteritis is related to 3 main factors: Viral enterotoxin (mainly the NSP4), malabsorption due to intestinal mucosal (absorptive enterocytes) damage and virus-induced down-regulation of the expression of absorptive enzymes, leading mainly to depletion of the enzyme ‘disaccharidases’. Although chemokine secreted by infected enterocytes plays an important active role in the pathogenesis [16] (Figure 7).
Immunity
The immune response to rotavirus infection is complex and not completely understood. Innate, cellular and humoral immunity contributes to eliminating the infection. Probably, antibodies against VP7 and VP4 are important for protection from the disease. This Humoral immunity has a dominant role to prevent severe disease on successive infection or on primary infection after immunization. Concerning Cell-mediated immunity, it probably plays a role in recovery from infection and in protection in addition to local immune factors such as secretory IgA or Interferon. Also, the duration of immunity against RV infection is limited and reinfection occurs in both children and adults but generally with less severity [17].
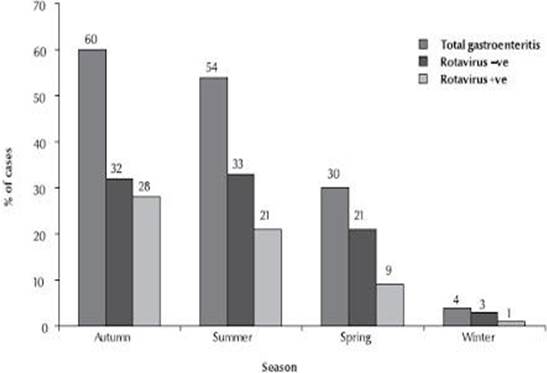
Figure 5: Seasonal Distribution of Rotavirus Infection. Adapted and Reprinted from Nafi O. Rotavirus Gastroenteritis among Children Aged Less Than 5 Years in Al Karak, Jordan. East Mediterr Health J. 2010 Oct; 16(10):1064-9. Copyright © World Health Organization (WHO), 2010
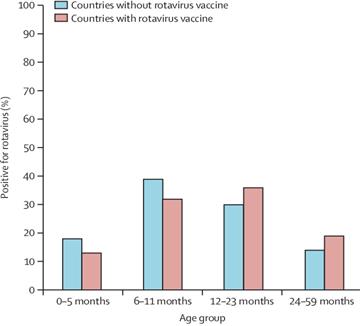
Figure 6: Age Distribution of Children Positive for Rotavirus in Countries with and without Rotavirus Vaccine Introduction. Adapted and Reprinted from Aliabadi N, Antoni S, Mwenda JM, Weldegebriel G, Biey JNM, Cheikh D, Fahmy K, Teleb N, Ashmony HA, Ahmed H, Daniels DS, Videbaek D, Wasley A, Singh S, de Oliveira LH, Rey-Benito G, Sanwogou NJ, Wijesinghe PR, Liyanage JBL, Nyambat B, Grabovac V, Heffelfinger JD, Fox K, Paladin FJ, Nakamura T, Agócs M, Murray J, Cherian T, Yen C, Parashar UD, Serhan F, Tate JE, Cohen AL. Global Impact of Rotavirus Vaccine Introduction on rotavirus Hospitalizations among Children under 5 years of age, 2008-16: Findings from the Global Rotavirus Surveillance Network. Lancet Glob Health. 2019 Jul; 7(7):e893-e903. Copyright © 2019 Published by Elsevier Ltd
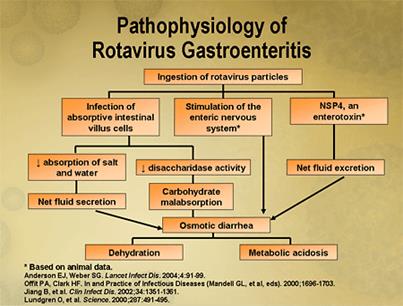
Figure 7: Rotavirus Pathogenesis. Adapted and Reprinted from Dr. Sherazi. Gastroenteritis. Available from Dr Sherazi (drsherazi110.blogspot.com)
Clinical Findings and Complications
RV is responsible for the major cause of diarrheal diseases in children below five years. The classical clinical features include watery diarrhea, fever, vomiting and abdominal pain. Infected children may also have a cough or runny nose. Also, RV infection may be asymptomatic, may cause self-limited watery diarrhea, or may result in dehydration (due to diarrhea, vomiting and fever) which may be severe and life threatening due to hypovolemia and circulatory collapse. In immunocompromised children, rotavirus infection can cause prolonged and chronic diarrhea. Generally, symptoms resolving occur between 3 to 7 days in mild and moderate infections. It has been reported that frequent RV infection has been associated with coeliac disease in genetically predisposed children [17] (Figure 8).
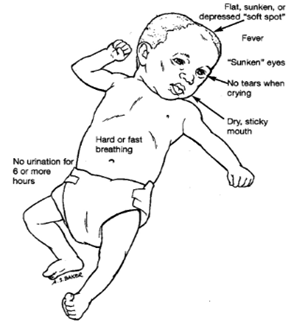
Figure 8: Signs of Dehydration. Adapted and Reprinted from Nationwide Children’s Hospital. Dehydration. Available from: Dehydration: Overview and Hydration Recommendations (nationwidechildrens.org)
Diagnosis
It is difficult to differentiate RV GE from other causes of GE by clinical spectrum alone. Also early diagnosis in hospitalized patients will decrease the morbidity and mortality considerably and avoid inappropriate use of antibiotics in pediatric patients. The most used method for diagnosis of RV infection is detection of rotavirus antigen in stool specimens by Enzyme ImmunoAssay (EIA) which is simple to use, inexpensive and with good sensitivity and specificity. RV antigen can be identified in the serum of patients 3–7 days after disease onset, but still the routine diagnostic testing is based primarily on testing of fecal specimens. In addition, the virus can be detected from stool by other techniques, used mainly in research settings such as Electron-microscopy, Immunoelectrophoresis, Flow cytometry, Latex Agglutination Test (LAT) and newly the Reverse Transcription Polymerase Chain Reaction (RT PCR), nucleic acid hybridization, Immunochromatography and more others [18].
Treatment
Since RV is stable in room temperature and highly contagious isolation of the patient is necessary. Treatment of RV GE is mainly by supportive measures, either by intravenous route or orally, in order to correct the loss of water and electrolytes that may lead to dehydration, acidosis, shock and death. In addition, symptomatic treatment of fever and vomiting is needed. Also, it is important to know that nor antibiotics or antivirals will help during a rotavirus infection [19]. The other key element for treatment of RV GE is nutritional support and early introduction to a normal diet. A Randomized controlled trial has demonstrated that other treatments such as probiotics, zinc, are effective in the management of acute gastroenteritis [18].
Preventive Measures
General Measures: As the main mode of transmission of Rotavirus is through faeco-oral route, proper hand hygiene, washing with soap and water after defecation and use of safe drinking water are mandatory. In addition, vitamin A supplementation is recommended in children since it reduces the incidence of diarrhea and associated deaths in children aged six months to five years [20].
Vaccine
Despite improvements in sanitation, limited effect on Rotavirus incidence is shown. So it is important to introduce safe and effective immunization to decrease the impact of RV infection in the community and reduce the worldwide burden of rotavirus disease. The first Rotavirus vaccine– ‘Rota-shield’ was introduced in United States (1998) by Wyeth and approved by Food and Drug Administration (USFDA) and Advisory Committee on Immunity Practices (ACIP) with an affectivity of 80-100% in preventing severe gastroenteritis caused by group A. However, one year later, the vaccine was removed from the market by the manufacturer since it was associated with increased risk of intussusception (1 in 12000 vaccinated infants) among vaccinated children [21]. Then, two new vaccines (Rotarix and RotaTeq) against Rotavirus Group A infection were introduced in 2006: 1. Rotarix® is an oral live attenuated vaccine containing the attenuated monovalent G1, P[8] human rotavirus strain and is recommended to be orally administered in two doses to infants at ages 2 months and 4 months.(or separated for at least 4 weeks), 2. RotaTeq® is a live attenuated, multivalent bovine-human reassortant rotavirus vaccine containing the most common rotavirus antigens seen in humans (G1, G2, G3, G4 and P[8]) and is recommended to be orally given in three doses, often at ages 2 months, 4 months and 6 months [22,23]. Therefore, WHO has recommended inclusion of RV vaccination in all national immunization programs? Then, the incidences and severity of Rotavirus infection has declined dramatically in countries that have adopted this recommendation and early estimates of the reduction in all-cause diarrhea hospitalizations in high-income countries have ranged from 20-50% and from 17 to 55% in middle-income countries. Also, the reduction in rotavirus hospitalizations has ranged from 49-92% in high-income countries, 54–59% in middle-income countries and 69–81% in low-income countries. These reductions have been sustained for several years after vaccine introduction [24]. In Lebanon, the Rotarix was introduced in late 2006 and RotaTeq was first available in 2009 but they have yet to be included in the National Immunization Program (NIP) [25] (Figure 9).
Returning to the risk of intussusception related to RV vaccines, a post licensure evaluation review (on 2012) concluded that Rotarix and RotaTeq were the effective vaccines and they were not associated with risk of intussusception in immunized children. The benefits of the vaccine far outweigh the small risk of intussusception [24].
Figure 9: Rotavirus Vaccine Administration. Adapted and Reprinted from Available from: UPSC Medical Sciences. Rotavirus Vaccine. Available from: Rotavirus Vaccine| UPSC Medico (medcampus.io)
In this context, it is important to mention that FDA recommends that the rotavirus vaccine should not be given to children who have a history of intussusception because they are more likely to have intussusception recurrence after receiving the RV vaccine. Additional RV vaccine contraindications include: Severe allergic reaction to a vaccine component (including latex) or following a prior dose of vaccine (latex rubber is contained in the RV1 oral applicator). Severe Combined Immunodeficiency (SCID). Finally, since the rotavirus vaccines became available, studies have shown that they prevent thousands of children from becoming sick and being hospitalized each year which highlight the importance of implantation of RV vaccine in the national immunization program in each country [26].
Objectives
Primary Objective: to evaluate the impact of vaccination program for rotavirus on disease severity among Lebanese infant’s population under 5 years of age who have been diagnosed with acute GE or RV GE from year 2016 to 2017 in 3 different hospitals localized in Beirut: RHUH, Al-Zahraa hospital and Sacré Coeur hospital.
Secondary Objectives
To estimate the rate of hospitalization related to RV in Lebanon, to determine the average age among RV associated hospitalizations and to compare the vaccine effectiveness between RV1 and RV5.