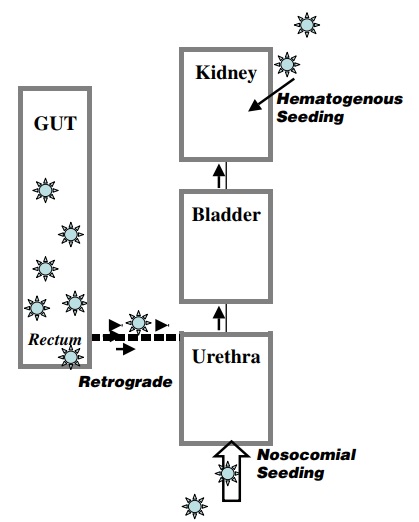
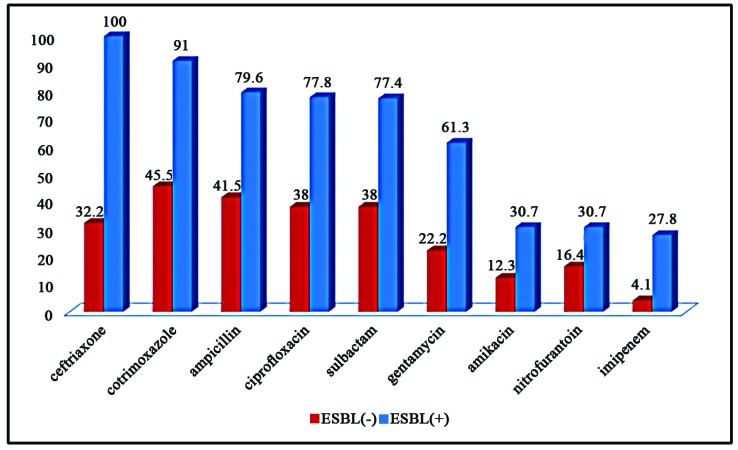
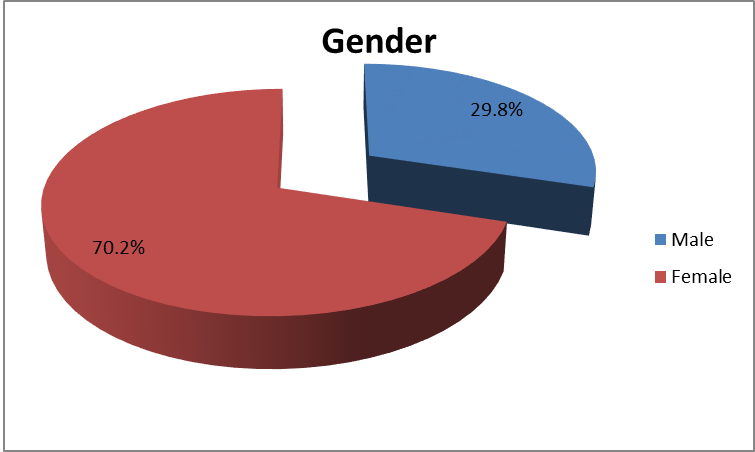
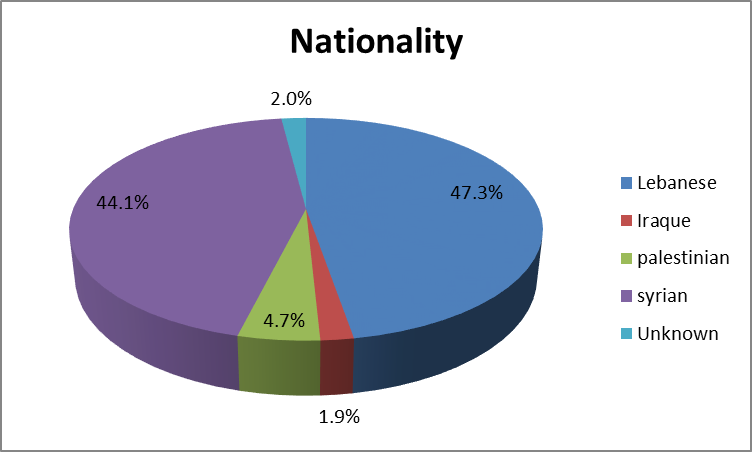
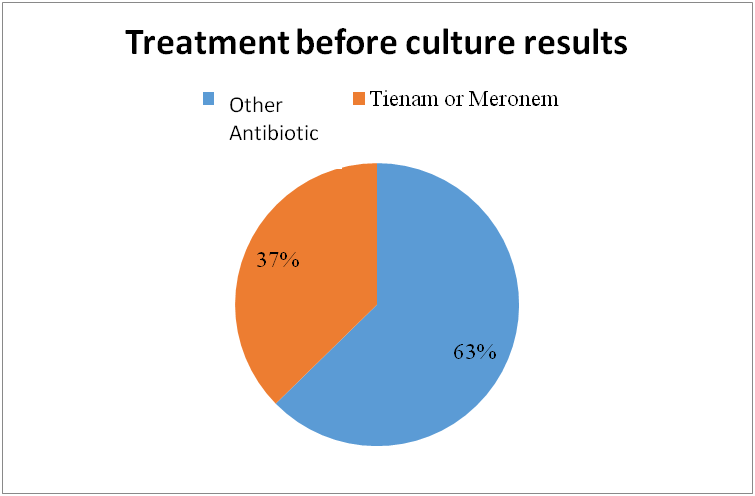
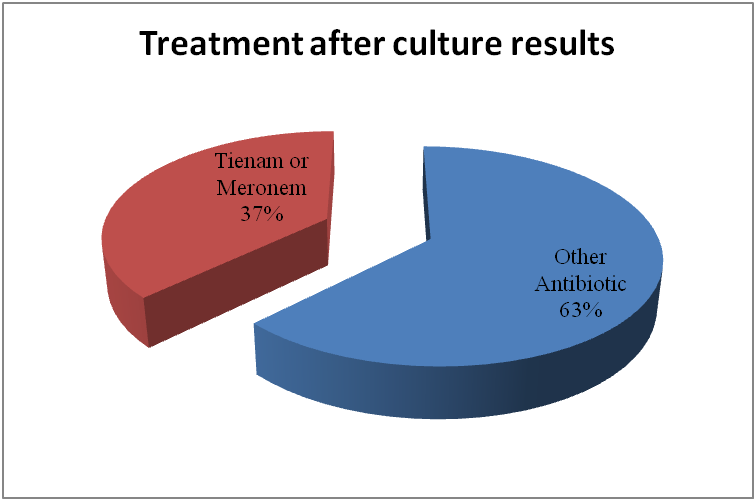
Leung, Alexander KC, et al. "Urinary tract infection in children." Recent patents on inflammation & allergy drug discovery 13.1 (2019): 2-18. doi: 10.2174/1872213X13666181228154940. PMID: 30592257. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6751349/
Bhat, Rahul G., Tamara A. Katy, and Frederick C. Place. "Pediatric urinary tract infections." Emergency Medicine Clinics 29.3 (2011): 637-653. doi: 10.1016/j.emc.2011.04.004. PMID: 21782079. https://linkinghub.elsevier.com/retrieve/pii/S0733-8627(11)00038-1
Korbel, Lindsey, Marianella Howell, and John David Spencer. "The clinical diagnosis and management of urinary tract infections in children and adolescents." Paediatrics and international child health 37.4 (2017): 273-279. doi: 10.1080/20469047.2017.1382046. PMID: 28978286. https://doi.org/10.1080/20469047.2017.1382046
Chang, Steven L., and Linda D. Shortliffe. "Pediatric urinary tract infections." Pediatric Clinics 53.3 (2006): 379-400. vi. doi: 10.1016/j.pcl.2006.02.011. PMID: 16716786. https://linkinghub.elsevier.com/retrieve/pii/S0031-3955(06)00027-7
Al-Mayahie, Sareaa, and Jaafar JH Al Kuriashy. "Distribution of ESBLs among Escherichia coli isolates from outpatients with recurrent UTIs and their antimicrobial resistance." The Journal of Infection in Developing Countries 10.06 (2016): 575-583. doi: 10.3855/jidc.6661. PMID: 27367005. https://jidc.org/index.php/journal/article/view/27367005
Becknell, Brian, et al. "The diagnosis, evaluation and treatment of acute and recurrent pediatric urinary tract infections." Expert review of anti-infective therapy 13.1 (2015): 81-90. doi: 10.1586/14787210.2015.986097. PMID: 25421102. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4652790/
White, Brett. "Diagnosis and treatment of urinary tract infections in children." American family physician 83.4 (2011): 409-415. PMID: 21322515. https://www.aafp.org/afp/2011/0215/p409.html
Wettergren, Bjorn, et al. "Six year follow up of infants with bacteriuria on screening." British Medical Journal 301.6756 (1990): 845-848. doi: 10.1136/bmj.301.6756.845. PMID: 2282422. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1663970/
Robinson, Joan L., et al. "Urinary tract infection in infants and children: Diagnosis and management." Paediatrics & child health 19.6 (2014): 315-319. PMID: 25332662. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4173959/
Prasad, Michaella Maloney, and Earl Y. Cheng. "Imaging studies and biomarkers to detect clinically meaningful vesicoureteral reflux." Investigative and clinical urology 58.Suppl 1 (2017): S23-S31. PMID: 28612057. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5468261/
Delbet, Jean Daniel, Mathie Lorrot, and Tim Ulinski. "An update on new antibiotic prophylaxis and treatment for urinary tract infections in children." Expert opinion on pharmacotherapy 18.15 (2017): 1619-1625. doi: 10.1080/14656566.2017.1383383. PMID: 28954556. https://www.tandfonline.com/doi/abs/10.1080/14656566.2017.1383383?journalCode=ieop20
Fernando, M. M. P. S. C., et al. "Extended spectrum beta lactamase producing organisms causing urinary tract infections in Sri Lanka and their antibiotic susceptibility pattern–a hospital based cross sectional study." BMC infectious diseases 17.1 (2017): 1-6. doi: 10.1186/s12879-017-2250-y. PMID: 28187754. https://pubmed.ncbi.nlm.nih.gov/28187754/
Hanna-Wakim, Rima H., et al. "Epidemiology and characteristics of urinary tract infections in children and adolescents." Frontiers in cellular and infection microbiology 5 (2015): 45. doi: 10.3389/fcimb.2015.00045. PMID: 26075187. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4443253/
Shaikh, Nader, et al. "Prevalence of urinary tract infection in childhood: a meta-analysis." The Pediatric infectious disease journal 27.4 (2008): 302-308. doi: 10.1097/INF.0b013e31815e4122. PMID: 18316994. https://journals.lww.com/pidj/Abstract/2008/04000/Prevalence_of_Urinary_Tract_Infection_in.4.aspx
Simões e Silva, Ana Cristina, and Eduardo Araújo Oliveira. "Update on the approach of urinary tract infection in childhood." Jornal de pediatria 91 (2015): S2-S10. doi: 10.1016/j.jped.2015.05.003. PMID: 26361319. https://www.sciencedirect.com/science/article/pii/S0021755715001199?via%3Dihub
Zorc, Joseph J., Darcie A. Kiddoo, and Kathy N. Shaw. "Diagnosis and management of pediatric urinary tract infections." Clinical microbiology reviews 18.2 (2005): 417-422. doi: 10.1128/CMR.18.2.417-422.2005. PMID: 15831830. https://pubmed.ncbi.nlm.nih.gov/15831830/
Hellström, A., et al. "Association between urinary symptoms at 7 years old and previous urinary tract infection." Archives of disease in childhood 66.2 (1991): 232-234. doi: 10.1136/adc.66.2.232. PMID: 2001110. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1792822/
Larcombe, James. "Urinary tract infection in children." Bmj 319.7218 (1999): 1173-1175. PMID: 21121537. https://www.aafp.org/afp/2010/1115/p1252.html
Zorc, Joseph J., et al. "Clinical and demographic factors associated with urinary tract infection in young febrile infants." Pediatrics 116.3 (2005): 644-648. doi: 10.1542/peds.2004-1825. PMID: 16140703. https://pediatrics.aappublications.org/content/116/3/644.long.
Chesney, Russell W., et al. "Randomized intervention for children with vesicoureteral reflux (RIVUR): background commentary of RIVUR investigators." Pediatrics 122.Supplement_5 (2008): S233-S239. doi: 10.1542/peds.2008-1285c. PMID: 19018047. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4336951/
Spencer, John David, et al. "Pediatric urinary tract infections: an analysis of hospitalizations, charges, and costs in the USA." Pediatric Nephrology 25 (2010): 2469-2475. doi: 10.1007/s00467-010-1625-8. PMID: 20711740. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4741383/
Spencer, John David, et al. "Pediatric urinary tract infections: an analysis of hospitalizations, charges, and costs in the USA." Pediatric Nephrology 25 (2010): 2469-2475. doi: 10.1016/j.ucl.2004.04.016. PMID: 15313061. https://doi.org/10.1016/j.ucl.2004.04.016
Robson, William Lane M., and Alexander KC Leung. "Explanation for false-positive urine cultures obtained by bag technique." Archives of pediatrics & adolescent medicine 149.9 (1995): 1042-1042. . doi: 10.1001/archpedi.1995.02170220107023. PMID: 7655595. https://jamanetwork.com/journals/jamapediatrics/article-abstract/517664
Morello, William, et al. "Acute pyelonephritis in children." Pediatric Nephrology 31 (2016): 1253-1265. doi: 10.1007/s00467-015-3168-5. PMID: 26238274. https://link.springer.com/article/10.1007%2Fs00467-015-3168-5
Chowdhury, Paramit, Steven H. Sacks, and Neil S. Sheerin. "Minireview: functions of the renal tract epithelium in coordinating the innate immune response to infection." Kidney international 66.4 (2004): 1334-1344. doi: 10.1111/j.1523-1755.2004.00896.x. PMID: 15458426. https://pubmed.ncbi.nlm.nih.gov/15458426/
Mulvey, Matthew A., et al. "Bad bugs and beleaguered bladders: interplay between uropathogenic Escherichia coli and innate host defenses." Proceedings of the National Academy of Sciences 97.16 (2000): 8829-8835. doi: 10.1073/pnas.97.16.8829. PMID: 10922042. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC34019/
Sobel, Jack D. "Pathogenesis of urinary tract infection: role of host defenses." Infectious disease clinics of North America 11.3 (1997): 531-549. https://doi.org/10.1016/S0891-5520(05)70372-X
Balighian, Eric, and Michael Burke. "Urinary tract infections in children." Pediatrics in review 39.1 (2018): 3-12. doi: 10.1542/pir.2017-0007. PMID: 29292282. https://pedsinreview.aappublications.org/content/39/1/3.long
Desai, Devang J., Brent Gilbert, and Craig A. McBride. "Paediatric urinary tract infections: Diagnosis and treatment." Australian family physician 45.8 (2016): 558-564. https://www.racgp.org.au/afp/2016/august/paediatric-urinary-tract-infections-diagnosis-and-treatment/
Hanson, LARS A. "Esch. coli infections in childhood. Significance of bacterial virulence and immune defence." Archives of disease in childhood 51.10 (1976): 737-742. doi: 10.1136/adc.51.10.737. PMID: 795381. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1546127/
Schlager, Theresa A. "Urinary tract infections in infants and children." Microbiology spectrum 4.5 (2016): 10-1128. doi: 10.1128/microbiolspec.UTI-0022-2016. PMID: 28087926. https://pubmed.ncbi.nlm.nih.gov/28087926/
Clark, Curtis J., William A. Kennedy, and Linda D. Shortliffe. "Urinary tract infection in children: when to worry." Urologic Clinics 37.2 (2010): 229-241. doi: 10.1016/j.ucl.2010.03.009. PMID: 20569801. https://www.sciencedirect.com/science/article/abs/pii/S0094014310000157?via%3Dihub
Karmazyn, Boaz K., et al. "ACR appropriateness criteria® urinary tract infection—child." Journal of the American College of Radiology 14.5 (2017): S362-S371. doi: 10.1016/j.jacr.2017.02.028. PMID: 28473093. https://linkinghub.elsevier.com/retrieve/pii/S1546-1440(17)30211-9
Mårild, Staffan, et al. "Protective effect of breastfeeding against urinary tract infection." Acta Paediatrica 93.2 (2004): 164-167. doi: 10.1080/08035250310007402. PMID: 15046267. https://doi.org/10.1111/j.1651-2227.2004.tb00699.x
Wiswell, Thomas E., Franklin R. Smith, and JAMES W. BASS. "Decreased incidence of urinary tract infections in circumcised male infants." Pediatrics 75.5 (1985): 901-903. PMID: 3991278. https://pediatrics.aappublications.org/content/75/5/901.long
To, Teresa, et al. "Cohort study on circumcision of newborn boys and subsequent risk of urinary-tract infection." The Lancet 352.9143 (1998): 1813-1816. doi: 10.1016/S0140-6736(98)02392-7. PMID: 9851381. https://pubmed.ncbi.nlm.nih.gov/9851381/
Schoen, Edgar J., Christopher J. Colby, and G. Thomas Ray. "Newborn circumcision decreases incidence and costs of urinary tract infections during the first year of life." Pediatrics 105.4 (2000): 789-793. doi: 10.1542/peds.105.4.789. PMID: 10742321. https://doi.org/10.1542/peds.105.4.789
Singh-Grewal, Davinder, Joseph Macdessi, and Jonathan Craig. "Circumcision for the prevention of urinary tract infection in boys: a systematic review of randomised trials and observational studies." Archives of Disease in Childhood 90.8 (2005): 853-858. doi: 10.1136/adc.2004.049353. PMID: 15890696. https://pubmed.ncbi.nlm.nih.gov/15890696/
Sampaio, Clara, et al. "Constipation and lower urinary tract dysfunction in children and adolescents: a population-based study." Frontiers in pediatrics 4 (2016): 101. doi: 10.3389/fped.2016.00101. PMID: 27752507. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5046079/
de Rovetto, Consuelo Restrepo. "Urinary tract infection: a prevalent problem in Pediatrics." Boletin medico del Hospital Infantil de Mexico 74.4 (2017): 241-242. doi: 10.1016/j.bmhimx.2017.06.001. PMID: 29382513. https://doi.org/10.1016/j.bmhimx.2017.06.001
Chase, Janet, et al. "The management of dysfunctional voiding in children: a report from the Standardisation Committee of the International Children's Continence Society." The Journal of urology 183.4 (2010): 1296-1302. doi: 10.1016/j.juro.2009.12.059. PMID: 20171678. https://www.auajournals.org/doi/10.1016/j.juro.2009.12.059
Keren, Ron, et al. "Risk factors for recurrent urinary tract infection and renal scarring." Pediatrics 136.1 (2015): e13-e21. doi: 10.1542/peds.2015-0409. PMID: 26055855. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4485012/
- AlKhater, Suzan A. "Approach to the child with recurrent infections." Journal of Family and Community Medicine 16.3 (2009): 77-82. PMID: 23012196. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3377046/
Yang, Jianhuan, et al. "Low serum 25-hydroxyvitamin D level and risk of urinary tract infection in infants." Medicine 95.27 (2016). doi: 10.1097/MD.0000000000004137. PMID: 27399128. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5058857/
Mahyar, Abolfazl, et al. "The role of overweight and obesity in urinary tract infection in children." Le infezioni in medicina 24.1 (2016): 38-42. PMID: 27031895. https://www.infezmed.it/index.php/article?Anno=2016&numero=1&ArticoloDaVisualizzare=Vol_24_1_2016_38
Filler, Guido, et al. "Prevention of chronic kidney disease in spina bifida." International urology and nephrology 44 (2012): 817-827. doi: 10.1007/s11255-010-9894-5. PMID: 21229390. https://link.springer.com/article/10.1007%2Fs11255-010-9894-5
Lo, Denise Swei, et al. "High frequency of Staphylococcus saprophyticus urinary tract infections among female adolescents." The Pediatric infectious disease journal 34.9 (2015): 1023-1025. doi: 10.1097/INF.0000000000000780. PMID: 26075812. https://journals.lww.com/pidj/Fulltext/2015/09000/High_Frequency_of_Staphylococcus_Saprophyticus.26.aspx
Al-Orifi, Fahad, et al. "Urine culture from bag specimens in young children: are the risks too high?." The Journal of pediatrics 137.2 (2000): 221-226. doi: 10.1067/mpd.2000.107466. PMID: 10931415. https://linkinghub.elsevier.com/retrieve/pii/S0022-3476(00)71927-5
Schmiemann, Guido, et al. "The diagnosis of urinary tract infection: a systematic review." Deutsches Ärzteblatt International 107.21 (2010): 361. doi: 10.3238/arztebl.2010.0361. PMID: 20539810. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2883276/
Garout, Wallaa A., et al. "Urinary tract infection in children younger than 5 years: etiology and associated urological anomalies." Saudi medical journal 36.4 (2015): 497. doi: 10.15537/smj.2015.4.10770. PMID: 25828291. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4404488/
Gounden, V., H. Bhatt, and I. Jialal. "Renal function tests.[Updated 2020 Jul 20]." StatPearls [Internet] (2020). Available from: https://www.ncbi.nlm.nih.gov/books/NBK507821/
Prasad, Michaella Maloney, and Earl Y. Cheng. "Imaging studies and biomarkers to detect clinically meaningful vesicoureteral reflux." Investigative and clinical urology 58.Suppl 1 (2017): S23-S31. PMID: 28612057. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5468261/
Shaikh, Nader, et al. "Early antibiotic treatment for pediatric febrile urinary tract infection and renal scarring." JAMA pediatrics 170.9 (2016): 848-854. doi: 10.1001/jamapediatrics.2016.1181. PMID: 27455161. https://jamanetwork.com/journals/jamapediatrics/fullarticle/2534481
Jayaweera, Jayaweera Arachchige Asela Sampath, and Mohommed Reyes. "RETRACTED ARTICLE: Antimicrobial misuse in pediatric urinary tract infections: recurrences and renal scarring." Annals of clinical microbiology and antimicrobials 17 (2018): 1-12. doi: 10.1186/s12941-018-0279-4. PMID: 29940982. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6016131/
Le Saux, Nicole, Canadian Paediatric Society, and Infectious Diseases and Immunization Committee. "Antimicrobial stewardship in daily practice: Managing an important resource." Paediatrics & child health 19.4 (2014): 261-265. . doi: 10.1093/pch/19.5.261. PMID: 24855430. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4029237/
Montini, Giovanni, Kjell Tullus, and Ian Hewitt. "Febrile urinary tract infections in children." New England Journal of Medicine 365.3 (2011): 239-250. doi: 10.1056/NEJMra1007755. PMID: 21774712. https://pubmed.ncbi.nlm.nih.gov/21774712/
Malande, Oliver Ombeva, et al. "A ten-year review of ESBL and non-ESBL Escherichia coli bloodstream infections among children at a tertiary referral hospital in South Africa." PloS one 14.9 (2019): e0222675. doi: 10.1371/journal.pone.0222675. PMID: 31550295. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6759190/
Yousefipour, Mehdi, et al. "Bacteria producing extended spectrum β-lactamases (ESBLs) in hospitalized patients: Prevalence, antimicrobial resistance pattern and its main determinants." Iranian journal of pathology 14.1 (2019): 61. doi: 10.30699/IJP.14.1.61. PMID: 31531102. https://pubmed.ncbi.nlm.nih.gov/31531102/
Frakking, Florine NJ, et al. "Appropriateness of empirical treatment and outcome in bacteremia caused by extended-spectrum-β-lactamase-producing bacteria." Antimicrobial agents and chemotherapy 57.7 (2013): 3092-3099. doi: 10.1128/AAC.01523-12. PMID: 23612198. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3697326/
Ruppé, Étienne, Paul-Louis Woerther, and François Barbier. "Mechanisms of antimicrobial resistance in Gram-negative bacilli." Annals of intensive care 5 (2015): 1-15. doi: 10.1186/s13613-015-0061-0. PMID: 26261001. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4531117/
Al-Assil, Bodour, Maysa Mahfoud, and Abdul Rezzak Hamzeh. "Resistance trends and risk factors of extended spectrum β-lactamases in Escherichia coli infections in Aleppo, Syria." American journal of infection control 41.7 (2013): 597-600. doi: 10.1016/j.ajic.2012.09.016. PMID: 23337303. https://www.ajicjournal.org/article/S0196-6553(12)01260-6/fulltext
Topaloglu, Rezan, et al. "Risk factors in community-acquired urinary tract infections caused by ESBL-producing bacteria in children." Pediatric nephrology 25 (2010): 919-925. doi: 10.1007/s00467-009-1431-3. PMID: 20151161. https://link.springer.com/article/10.1007%2Fs00467-009-1431-3
Republic of Lebanon Ministry of Public Health. (2016). Syrian Refugees Crisis Impact on Lebanese Public Hospitals- Financial Impact Analysis: APIS Report. 2016. Accessed on December 22, 2020. Retrieved from: https://www.moph.gov.lb/en/Pages/127/9287/syrian-refugees-crisis-impact-on-lebanese-public-hospitals-financial-impact-analysis-apis-report
Al-Ibrahim, Alia Abdulrahim, et al. "Urinary tract infection and vesicoureteral reflux in Saudi children." Saudi Journal of Kidney Diseases and Transplantation 13.1 (2002): 24-28. . PMID: 18209408.
Milovanovic, Tamara, et al. "Epidemiology and risk factors for multi-drug resistant hospital-acquired urinary tract infection in patients with liver cirrhosis: single center experience in Serbia." BMC infectious diseases 19.1 (2019): 1-10. doi: 10.1186/s12879-019-3761-5. PMID: 30755176. https://doi.org/10.1186/s12879-019-3761-5
Bitsori, Maria, and Emmanouil Galanakis. "Pediatric urinary tract infections: diagnosis and treatment." Expert review of anti-infective therapy 10.10 (2012): 1153-1164. doi: 10.1586/eri.12.99. PMID: 23199401. https://doi.org/10.1586/eri.12.99
Hsiao, Chih-Yen, et al. "Risk factors for development of acute kidney injury in patients with urinary tract infection." PloS one 10.7 (2015): e0133835. doi: 10.1371/journal.pone.0133835. PMID: 26213991. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4516244/
Salem, S. E., Elias Dahdouh, and Ziad Daoud. "Resistance of gram-negative bacilli in Lebanon." International Scholarly Research Notices 2013 (2013). doi: 10.5402/2013/759208. https://doi.org/10.5402/2013/759208
Garrido, David, et al. "Clinical characterization and antimicrobial resistance of Escherichia coli in pediatric patients with urinary tract infection at a third level hospital of Quito, Ecuador." Boletin Medico Del Hospital Infantil de Mexico 74.4 (2017): 265-271. doi: 10.1016/j.bmhimx.2017.02.004. PMID: 29382515. https://linkinghub.elsevier.com/retrieve/pii/S1665-1146(16)30138-1
Flokas, Myrto Eleni, et al. "Prevalence of ESBL-producing Enterobacteriaceae in paediatric urinary tract infections: a systematic review and meta-analysis." Journal of Infection 73.6 (2016): 547-557. doi: 10.1016/j.jinf.2016.07.014. PMID: 27475789. https://linkinghub.elsevier.com/retrieve/pii/S0163-4453(16)30178-5
Paterson, D. L. "Recommendation for treatment of severe infections caused by Enterobacteriaceae producing extended-spectrum β-lactamases (ESBLs)." Clinical Microbiology and Infection 6.9 (2000): 460-463. doi: 10.1046/j.1469-0691.2000.00107.x. PMID: 11168179. https://linkinghub.elsevier.com/retrieve/pii/S1198-743X(14)63150-5